| Name | Joyce Jean Lu, Ph.D. |
|---|---|
| Position | Associate Professor |
| 所屬單位 |
stem cell renewal, de-differentiation, reprogramming, differentiation, embryonic stem cells, mesenchymal stem cells, totipotent, retinal progenitor cells. high-throughput functional screen Ph.D. Institute of Microbiology, National Taiwan University, Taipei, Taiwan. 1995-2000 M.S. Institute of Molecular Medicine, National Taiwan University, Taipei, Taiwan. 1992-1994 B.S. Department of Medical Technology, National Taiwan University, Taipei, Taiwan. 1988-1992 |
| 名片 | In Memory of Dr. Joyce Jean Lu |
| 研究方向/領域 |
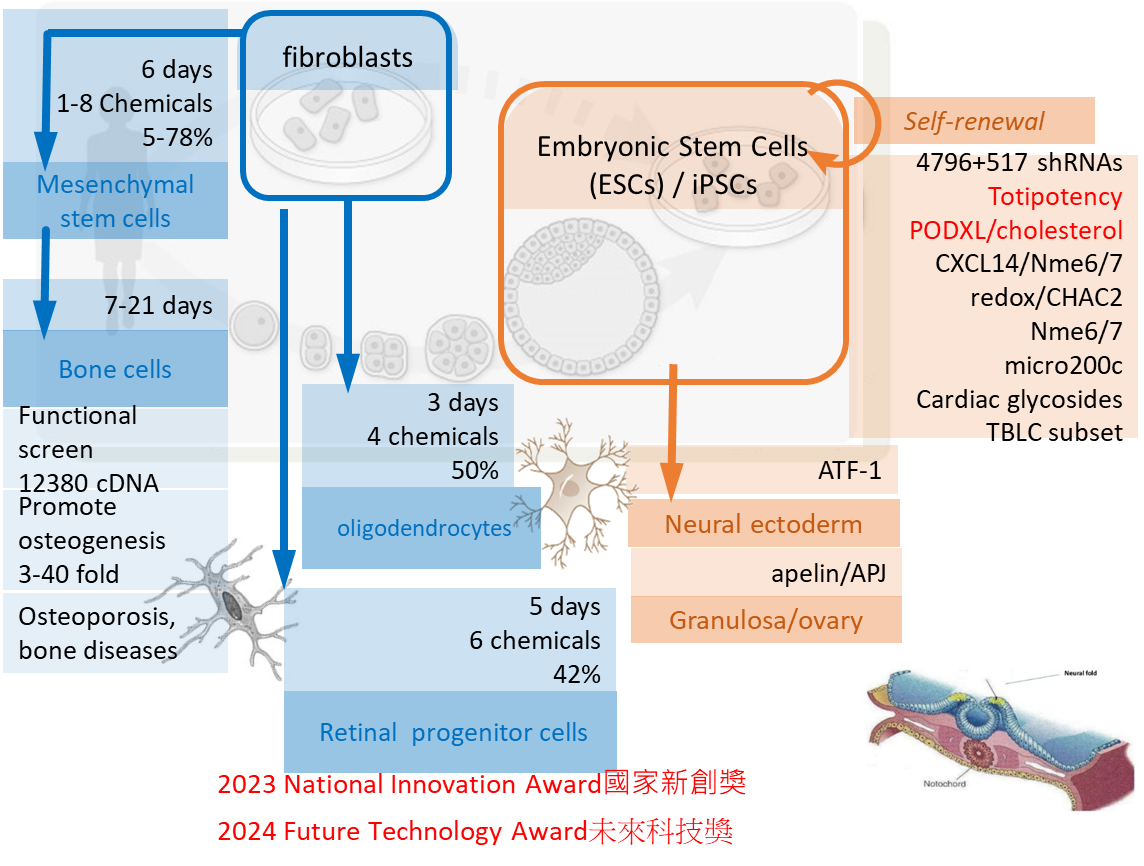
RESEARCH INTERESTSStem cell renewal/de-differentiation/differentiation is important for stem cell biology and regenerative medicine. To efficiently pinpoint factors critical for cell fate determination, systematic functional screens were performed in mouse embryonic stem cells (ESCs), human ESCs, and mesenchymal stem cells (MSCs). We established the first shRNA screen in ESCs. With a shRNA library of kinases and phosphatases containing 4796 shRNAs, we identified 244 genes essential for ESC renewal. Among them, Nme6 and Nme7 are necessary for 8 stemness gene expressions. With 517 shRNA target genes differentially expressed in undifferentiated human ESCs, we performed a shRNA functional screen and found 86 genes essential for human ESC renewal. Two candidate genes, either bridging the oxidation and reduction status of ESCs with stemness signals, or blocking neural stem cell differentiation, are currently being studied in-depth. To fulfill the unmet need for bone diseases, a gain-of-function screen was performed with an overexpression library containing 12380 genes. 9 candidate genes are found to be essential and sufficient to promote the osteogenesis of human MSCs. By this method, we found soluble factors that can completely treat and prevent osteoporosis. In addition, we found PODXL/cholesterol pathway can promote human mouse chimerism by up to 57% and published in Advanced Science (impact factor 17.521). Recently, how to dedifferentiate/reprogram stem cells into totipotency that can differentiate stem cells into all cell types has become an important topic. We develop a novel cocktail that can uniquely trigger the reprogramming of totipotent stem cells.We also differentiate fibroblasts into mesenchymal stem cells, oligodendrocytes, and retinal progenitor cells. We won the 2024 Future Technology Award and the 2023 National Innovation Award. Thus, we found many factors play critical roles in ESC/ MSC/fibroblast renewal, de-differentiation, and differentiation by systematically functional screens. (edited by ChatGPT)
|
EDUCATION AND POSITIONS HELD
- 2018-2025 Adjunct Associate Professor, Graduate Institute of Medical Sciences, National Defense Medical Center, Taipei, Taiwan
- 2016-2025 Adjunct Associate Professor, Department of Life Science, Tzu Chi University, Hualien, Taiwan
- 2015-2025 Associate Research Fellow, Genomics Research Center, Academia Sinica, Taiwan
- 2015-2025 Adjunct Associate Professor, Genomics and System Biology Program, College of Life Science, National Taiwan University, Taiwan
- 2009-2023 Co-PI, National RNAicore Facility Plateform
- 2007-2015 Assistant Research Fellow, Stem Cell Program, Genomics Research Center, Academia Sinica, Taiwan
- 2010-2015 Adjunct Assistant Professor, Genomics and System Biology Program, College of Life Science, National Taiwan University, Taiwan
- 2003-2007 Postdoctoral Fellow/Associate, Molecular, Department of Cellular, and Developmental Biology, Yale University, USA
- 2001-2003 Postdoctoral Fellow, Graduate Institute of Microbiology, College of Medicine, National Taiwan University, Taiwan
- 1994-1995 Research Assistant, Graduate Institute of Microbiology, College of Medicine, National Taiwan University, Taiwan
HONORS
- 2024 Future Technology Award
- 2023 National Innovation Award
- 2017 Keystone symposia scholarship. Regeneration Biology and applications: cell differentiation, tissue organization and biomedical engineering.
- 2003-2005 Ruth L. Kirschstein National Research Service Award Fellowship, Ruth L. Kirschstein
- 2002 Outstanding Paper Award (National Taiwan University)
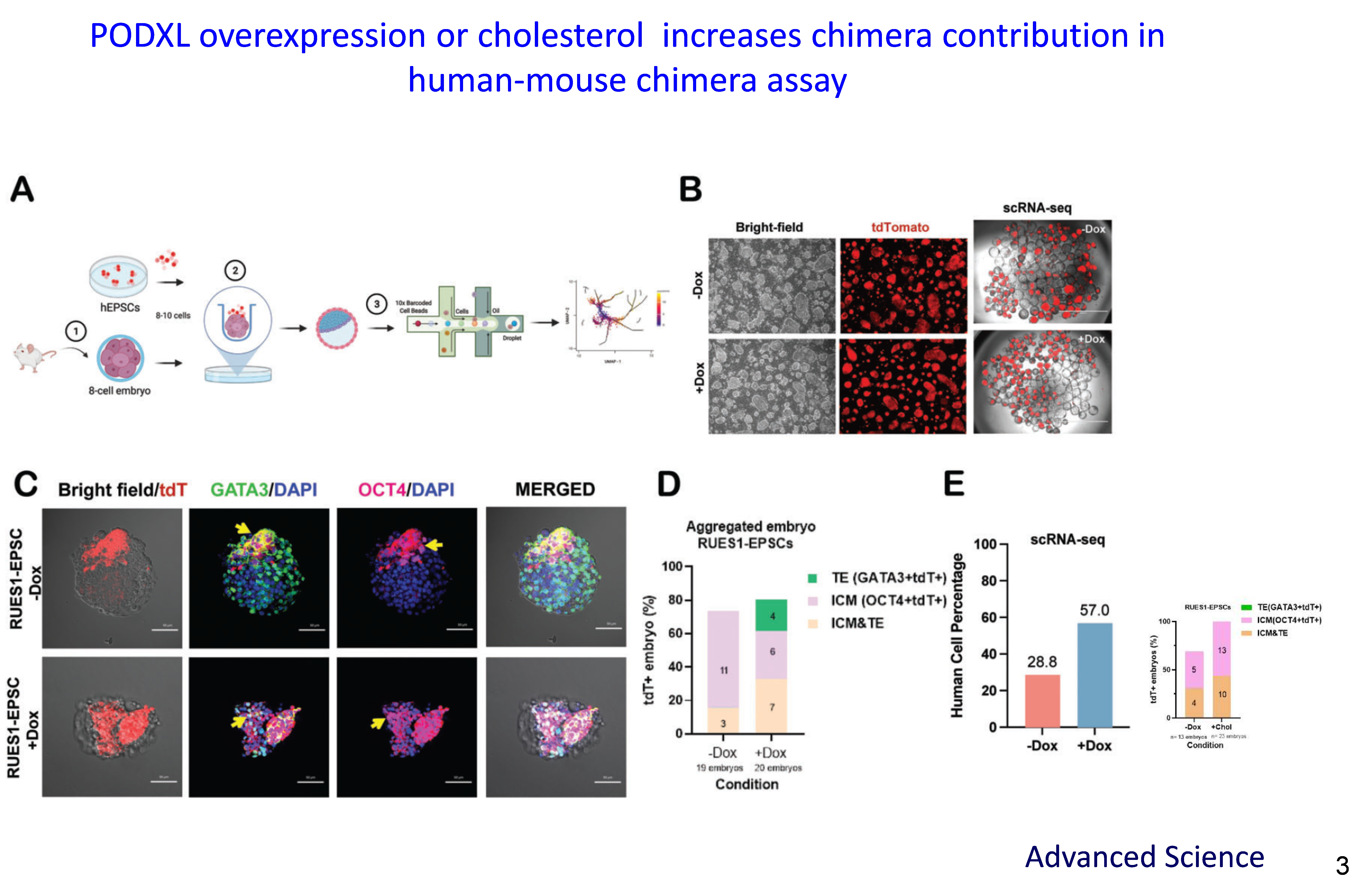
We uncovered Podocalyxin-like protein 1 (PODXL) /cholesterol signals critical for maintaining the extended pluripotent stem cell (EPSC) pluripotent state, which is essential for understanding early embryonic development. Altering PODXL expression levels affects self-renewal capacity, protein expression of c-MYC/ telomerase, and colony formation efficiency of induced pluripotent stem cells (iPSCs) and EPSCs.
We are the first group to report that PODXL functions as a membrane protein regulator of cholesterol biosynthesis. Human pluripotent stem cells (hPSCs) exhibit greater sensitivity to cholesterol depletion compared to fibroblasts. The addition of exogenous cholesterol fully rescued the loss of pluripotency caused by PODXL gene silencing. PODXL influences lipid raft dynamics by modulating intracellular cholesterol levels. Specifically, PODXL regulates the recruitment of the RAC1/CDC42 network to control the maturation of SREBP1 and SREBP2, thereby affecting lipid raft dynamics.
Single-cell RNA sequencing further revealed that PODXL overexpression, along with cholesterol supplementation, markedly enhanced the contribution of human cells within human-mouse chimeric embryos, achieving a chimerism rate of 57%. These findings underscore the pivotal role of PODXL-mediated cholesterol signaling in regulating pluripotency in ESCs and EPSCs.
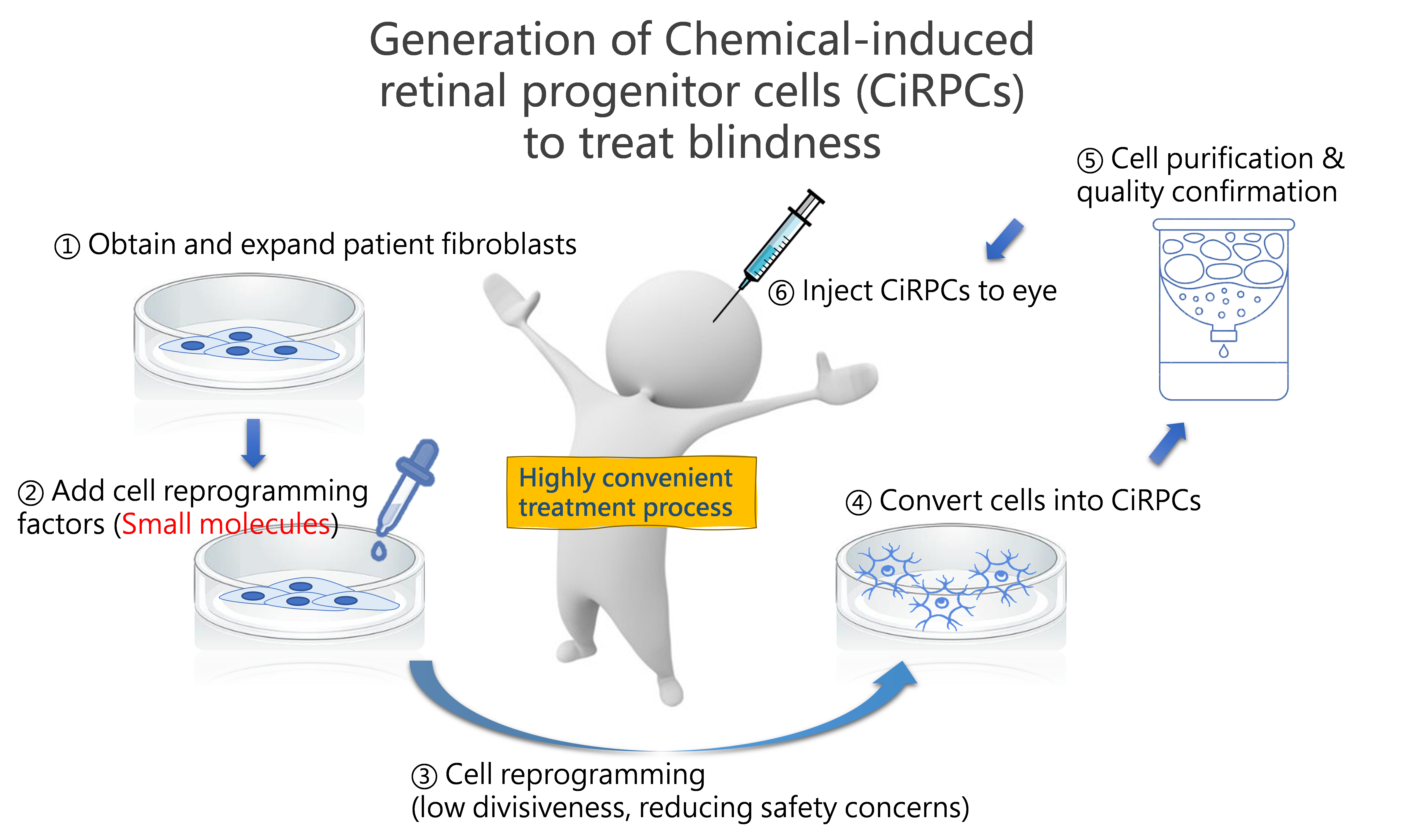
Direct Reprogramming of Human Fibroblasts into Retinal Progenitor Cells Using Small Molecules for Photoreceptor Degeneration Therapy
We successfully reprogrammed human fibroblasts into retinal progenitor cells (RPCs) using a small-molecule-based approach, avoiding traditional gene editing or viral transduction techniques. This innovative method has demonstrated outstanding therapeutic efficacy in animal models without the risk of tumorigenesis.
The reprogramming protocol is straightforward, cost-effective, and exhibits high conversion efficiency, offering new hope for patients suffering from vision loss. The breakthrough work on RPCs highlights their potential in regenerative ophthalmology.
Looking ahead, the team aims to establish partnerships with medical institutions and pharmaceutical companies to accelerate the clinical translation of RPC-based cell therapies. This significant advancement marks a promising step toward restoring sight and improving the quality of life for individuals affected by photoreceptor degeneration like retinitis pigmentosa (orphan drug), macular degeneration, diabetic retinopathy.