 研究員
研究員
cma
02-27871233
02-27871233
EDUCATION AND POSITIONS HELD:
- B.S., Chemistry, National Taiwan University, 1992
- M.S., Chemistry, University of Pennsylvania, 1996
- Ph.D., Chemistry, University of Pennsylvania, 2000
- Postdoctoral Fellow, University of California at San Diego, 2001
- Postdoctoral Fellow, The Scripps Research Institute, 2001–2004
- Assistant Professor, Genomics Research Center, Academia Sinica, Taiwan, 2004-2010
- Associate Professor, Genomics Research Center, Academia Sinica, Taiwan, 2010-2019
- Visiting Scientist, RIKEN SPring-8 Center, Japan, 2016 – present
- Division Director of Chemical Biology, GRC, Academia Sinica, Taiwan, 2016 – 2023
- Professor, Genomics Research Center, Academia Sinica, Taiwan, 2019-present
HONORS:
- University of Pennsylvania, Teaching Assistant Award, 1995
- Keystone Symposia Scholarship, Frontiers of Structural Biology, 2003
- The Skaggs Postdoctoral Fellowship, 2001–2003
- TWAS Young Affiliate, 2009-2013
- Academia Sinica Significant Research Achievements, 2009
- Academia Sinica Research Award for Junior Research Investigators, 2010
- Two research highlights in Taiwan Yearbook of Technology, 2010
- The Young Scholar Award of Tien-De Li Biomedical Foundation, 2011
- Academia Sinica Significant Research, 2012
- Academia Sinica Career Development Award, 2013
- Exceptional Merit in Academic Award from Chung Hwa Rotary Educational Foundation, 2014
- Taiwan Bio-Development Foundation Chair in Biotechnology, 2014
- Two FUTEX Futuretech Awards, Ministry of Science and Technology, Taiwan
RESEARCH INTERESTS:
Structure and functions of membrane glycoproteins in drug discovery
- In order to overcome the problems of drug-resistant bacterial infection, the membrane-bound bifunctional transglycosylase, has been chosen for structural and functional analysis. We have determined the X-ray crystal structures of this membrane-bound enzyme in complex with its inhibitor moenomycin, and with its substrate lipid II analog, leading to the proposed mechanism of peptidoglycan synthesis. In addition, the crystal structure of a membrane protein complex, the key component of divisome ftsBLQ has been determined to decipher the cell division process.
- Targeting the bacterial transglycosylase: antibiotic development from a structural perspective, X Chen, CH Wong, C Ma*. ACS Infectious Diseases, 5, 1493 (2019). (Times cited: 16)
- Structure of the heterotrimeric membrane protein complex FtsB-FtsL-FtsQ of the bacterial divisome. HTV Nguyen, X Chen, C Parada, AC Luo, O Shih, US Jeng, CY Huang, YL Shih* and C Ma*. Nat Commun. 14(1):1903 (2023).
- We have discovered a simple and practical approach using traditional egg-based method to prepare a broad-spectrum split influenza virus vaccine that when vaccinated can induce cross-strain protection in mice.
- Egg-based influenza split virus vaccine with monoglycosylation induces cross-strain protection against influenza virus infections, YC Tseng, CY Wu, ML Liu, TH Chen, WL Chiang, YH Yu, JT Jan, KI Lin, CH Wong*, C Ma*. Proc Natl Acad Sci U S A, 116, 4200 (2019). (Times cited: 28)
- Better influenza vaccines: an industry perspective. JR Chen, YM Liu, YC Tseng, C Ma*. J Biomed. Sci., 27, 33 (2020). (Times cited: 66)
- Chimeric hemagglutinin vaccine elicits broadly protective CD4 and CD8 T cell responses against multiple influenza strains and subtypes. HY Liao, SC Wang, YA Ko, KI Lin, C Ma, TJR Cheng, CH Wong. Proc Natl Acad Sci U S A, 117, 17757 (2020). (Times cited: 18)
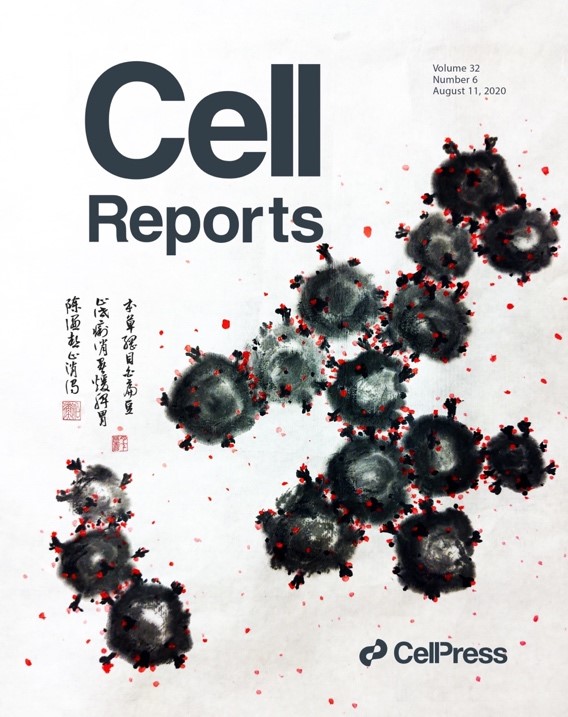
- We isolated a plant lectin FRIL from the edible Lablab beans with superior anti-influenza and anti-SARS-CoV-2 activities. FRIL binds preferentially to complex-type N-glycans and neutralizes viruses that possess complex-type N-glycans on their envelopes. As a homotetramer, FRIL is capable of aggregating influenza particles through multivalent binding and trapping influenza virions in cytoplasmic late endosomes, preventing their nuclear entry. Remarkably, FRIL also effectively neutralizes SARS-CoV-2, preventing viral protein production and cytopathic effect in host cells. These findings suggest a potential application of FRIL for the prevention and/or treatment of influenza and COVID-19.
- A carbohydrate-binding protein from the edible Lablab beans effectively blocks the infections of influenza viruses and SARS-CoV-2. YM Liu, M. Shahed-Al-Mahmud, X Chen, TH Chen, KS Liao, JM Lo, YM Wu, MC Ho, CY Wu, CH Wong, JT Jan, C Ma*. Cell Reports, 32, 108016 (2020), selected as journal cover. (Times cited: 62)
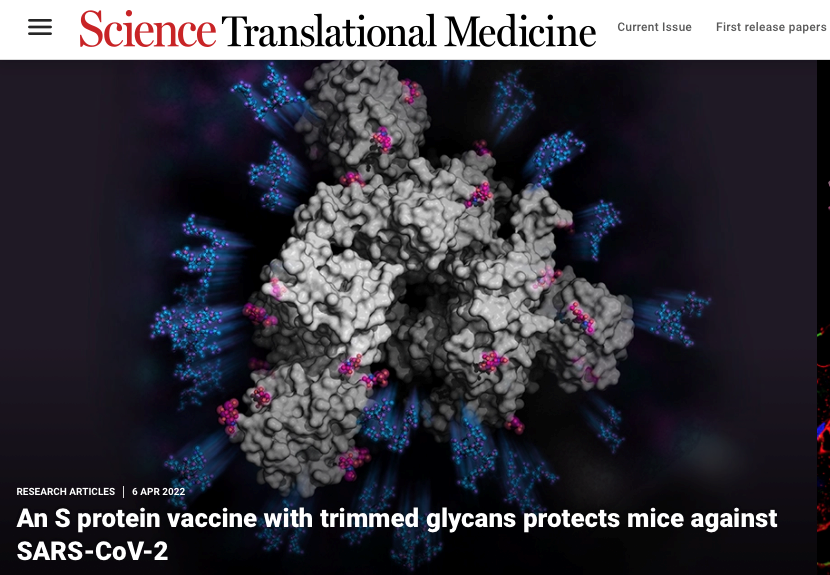
- We have studied the effect of glycosylation on SARS-CoV-2 virus major surface Spike protein with regards to its role in ACE2 receptor binding and immune response, and developed a simple strategy for broad-spectrum COVID-19 vaccine design. The resulting mono-GlcNAc-decorated Spike vaccination can elicit superior B cell, Tfh cell and CD8 T cell responses, and protect hACE2 transgenic mice against infections of SARS-CoV-2 and the variants of concerns, such as the original Wuhan, Alpha, Beta, Gamma, Delta and more recently Omicron and its subvariants. Antibody responses from a structural perspective are also been studied with samples from COVID-19 vaccination and convalescent individuals. Several structures have been determined to understand the structural basis of anti-RBD neutralizing antibodies.
- Vaccination with SARS-CoV-2 spike protein lacking glycan shields elicits enhanced protective responses in animal models. HY Huang, HY Liao, X Chen, SW Wang, CW Cheng, M Shahed-Al-Mahmud, YM Liu, A Mohapatra, TH Chen, JM Lo, YM Wu, HH Ma, YH Chang, HY Tsai, YC Chou, YP Hsueh, CY Tsai, PY Huang, SY Chang, TL Chao, HC Kao, YM Tsai, YH Chen, CY Wu, JT Jan, TJR Cheng, KI Lin*, C Ma* and CH Wong*. Science Translational Medicine, 14, eabm0899 (2022), selected as online feature article. (Times cited: 33)
- Glycosite-deleted mRNA of SARS-CoV-2 spike protein as a broad-spectrum vaccine. CY Wu, CW Cheng, CC Kung, KS Liao, JT Jan, C Ma and CH Wong*. Proc Natl Acad Sci U S A, 119, e2019995119 (2022). (Times cited: 13)
- Structures and therapeutic potential of anti-RBD human monoclonal antibodies against SARS-CoV-2. KYA Huang, D Zhou, TK Tan, C Chen, HME Duyvesteyn, Y Zhao, HM Ginn, L Qin, P Rijal, L Schimanski, R Donat, A Harding, J Gilbert-Jaramillo, W James, JA Tree, K Buttigieg, M Carroll, S Charlton, CE Lien, MY Lin, CP Chen, SH Cheng, X Chen, TY Lin, EE Fry, J Ren, C Ma, AR Townsend, DI Stuart. Theranostics, 12, 1 (2022). (Times cited: 5)
- Breadth and function of antibody response to acute SARS-CoV-2 infection in humans. KYA Huang, TK Tan, TH Chen, CG Huang, R Harvey, S Hussain, CP Chen, A Harding, J Gilbert-Jaramillo, X Liu, M Knight, L Schimanski, SR Shih, YC Lin, CY Cheng, SH Cheng, YC Huang, TY Lin, JT Jan, C Ma, W James, RS Daniels, Rodney JW McCauley, P Rijal, Pramila, AR Townsend. PLOS Pathogens, 17, e1009352 (2021). (Times cited: 55)
- Structural basis for a conserved neutralization epitope on the receptor-binding domain of SARS-CoV-2. KA Huang*, X Chen, A Mohapatra, HTV Nguyen, L Schimanski, TK Tan, P Rijal, SK Vester, RA Hills, M Howarth, JR Keeffe, AA Cohen, LM Kakutani, YM Wu, M Shahed-Al-Mahmud, YC Chou, PJ Bjorkman, AR Townsend, C Ma*. Nat Commun. 14(1):311 (2023).
- We have generated a monoclonal antibody to target an important membrane receptor IL-17RB for pancreatic cancer, which has an extremely high mortality rate due to its aggressive metastatic nature. Resolving the underlying mechanisms will be crucial for treatment. We found that overexpression of IL-17B receptor (IL-17RB) strongly correlated with postoperative metastasis and inversely correlated with progression-free survival in pancreatic cancer patients.
- Targeting IL-17B-IL-17RB signaling with an anti-IL-17RB antibody blocks pancreatic cancer metastasis by silencing multiple chemokines, HH Wu, WW Hwang-Verslues, WH Lee, CK Huang, PC Wei, CL Chen, JY Shew, EY Lee, YM Jeng, YW Tien, C Ma, WH Lee*, J Exp Medicine, 212, 333-349 (2015).
- Structural basis of interleukin-17B receptor in complex with a neutralizing antibody for guiding humanization and affinity maturation. WH Lee, X Chen, IJ Liu, JH Lee, CM Hu, HC Wu, SK Wang, WH Lee* and C Ma*. Cell Reports 41(4):111555 (2022).
- In August 2022, the technologies related to COVID-19 vaccine and antibodies have been transferred to a new startup biotech company by Academia Sinica for developing products such as broad-spectrum vaccines and antibodies with glycoengineering technologies.
膜蛋白及醣蛋白的結構生物學與新藥研發
馬徹實驗室的主要研究重點為以結構生物學探討新興感染疾病及癌症的重要課題,如疫苗設計,抗體及開發新型抗生素。特別是最近年以來對於新冠肺炎病毒的預防與治療,設計新型抗生素藥物等有三項重要貢獻:
一、發現去醣化棘蛋白疫苗能夠誘發針對新冠變種病毒的廣效保護。新冠肺炎病毒表面的主要抗原棘蛋白突變能力很強,而不變的抗原決定區通常被多醣所覆蓋。藉由醣蛋白工程去除棘突蛋白表面干擾免疫辨識的多醣,而製成去醣化棘突蛋白疫苗(SMG)可以誘發針對變異株的更佳的B細胞及T細胞反應,具有潛力成為次世代廣效型新冠肺炎疫苗,另外也以X光結晶學與冷凍電顯等技術,解構出新冠病毒表面上棘狀蛋白的中和抗體結合熱區;
二、發現食用白扁豆萃取物中的一個凝集素(FRIL)可以有效中和各種流感病毒以及各種冠狀病毒。此凝集素與病毒表面的複合型多醣有強烈的結合能力,使病毒無法進入細胞完成複製,進而抑制病毒入侵,有效阻斷病毒傳播。於小鼠實驗驗證,鼻腔吸入此凝集素可以有效保護高致命量之流感病毒感染,並提升存活率;
三、以X光結晶學方法解出細胞壁於分裂時負責調控的膜蛋白FtsBLQ複合體的結構,作為以分裂體為新抗生素標靶的基礎。
RESEARCH HIGHLIGHTS:
- 2023/04/07, 新興抗生素標靶 膜蛋白FtsBLQ結構與機轉模型首度曝光
- 2023/01/30, 預防變異 解析新冠病毒棘狀蛋白上中和抗體結合熱區又一突破
- 2022/10/26, 首揭IL-17RB抗原晶體結構 單株抗體邁向人類化
- 2022/03/03, 迎戰新冠變種病毒 去醣化棘突蛋白疫苗提供更有效的保護
- 2022/02/15, 終結新冠疫情新曙光? 廣效新冠肺炎RNA疫苗
- 2021/10/01, 科學人特別報導: 單醣化,力抗新冠病毒變異株
- 2021/01/27, 從現有藥物和保健品找出有效對抗新冠病毒感染的藥方
- 2020/11/03, 研之有物:未來流感疫苗不用年年打?可對抗異株病毒的單醣化疫苗
- 2020/08/11, 嵌合並單醣化血凝集素,做出可抗人及禽流感的廣效疫苗
- 2020/07/29, 白扁豆萃取蛋白可阻斷新冠病毒入侵
- 2019/02/20, 傳統方法製作單醣流感疫苗,抗流感不再有疑慮
- 2014/01/23, 廣效流感疫苗的新發展
- 2012/04/17, 馬徹實驗室發表細菌轉醣酶的完整解剖圖
- 2009/10/15, 恭賀馬徹助研究員獲選為TWAS年輕學者
- 2009/10/11, 流感病毒的感染以及疫苗設計的關鍵,都在於『醣』的多寡!
- 2009/08/25, 細菌膜蛋白酵素完整結構研究備受矚目
- 2009/05/21, 馬徹實驗室發表細菌膜蛋白酵素完整結構,尋找新型抗生素有譜
- 2008/01/14, 新突破方法開發新抗生素
SELECTED PUBLICATIONS:
- Kung CC, Lo JM., Liao KS, Wu CY, Cheng LC, Chung C, Hsu TL, Ma C*, Wong CH*, 2025, “Expression of Human β3GalT5–1 in Insect Cells as Active Glycoforms for the Efficient Synthesis of Cancer-Associated Globo-Series Glycans”, Journal of the American Chemical Society, 147(13), 10864-10874. (SCIE) (IF: 15; SCI ranking: 9.6%)
- Lo JM., Kung CC, Cheng TJR, Wong CH*, Ma C*, 2025, “Structure-Based Mechanism and Specificity of Human Galactosyltransferase β3GalT5”, Journal of the American Chemical Society, 147(13), 10875-10885. (SCIE) (IF: 15; SCI ranking: 9.6%)
- Chen X, Mohapatra A, Nguyen HTV, Schimanski L, Kit TT, Rijal P, Chen CP, Cheng SH, Lee WH, Chou YC, Townsend AR, Ma C, Huang KYA, 2024, “The presence of broadly neutralizing anti-SARS-CoV-2 RBD antibodies elicited by primary series and booster dose of COVID-19 vaccine”, PLOS Pathogens, 20(6), e1012246. (SCIE)
- Huang KA*, Chen X, Mohapatra A, Nguyen HTV, Schimanski L, Tan TK, Rijal P, Vester SK, Hills RA, Howarth M, Keeffe JR, Cohen AA, Kakutani LM, Wu YM, Shahed-Al-Mahmud M, Chou YC, Bjorkman PJ, Townsend AR, Ma C*, 2023, “Structural basis for a conserved neutralization epitope on the receptor-binding domain of SARS-CoV-2.”, Nature communications, 14(1), 311. (SCIE)
- Nguyen HTV, Chen X, Parada C, Luo AC, Shih O, Jeng US, Huang CY, Shih YL*, Ma C*, 2023, “Structure of the heterotrimeric membrane protein complex FtsB-FtsL-FtsQ of the bacterial divisome”, Nature Communications, 14(1), 1903. (SCIE)
- T Yong, KK Chang, YS Wang and C Ma*, 2022, “Active humoral response reverts tumorigenicity through disruption of key signaling pathway”, Vaccines, 10, 163. (SCIE)
- Daniell H, Nair SK, Guan H, Guo Y, Kulchar RJ, Torres MDT, Shahed-Al-Mahmud M, Wakade G, Liu YM, Marques AD, Graham-Wooten J, Zhou W, Wang P, Molugu SK, de Araujo WR, de la Fuente-Nunez C, Ma C, Short WR, Tebas P, Margulies KB, Bushman FD, Mante FK, Ricciardi RP, Collman RG, Wolff MS, 2022, “Debulking different Corona (SARS-CoV-2 delta, omicron, OC43) and Influenza (H1N1, H3N2) virus strains by plant viral trap proteins in chewing gums to decrease infection and transmission.”, Biomaterials, 288, 121671. (SCIE)
- Wu CY, Cheng CW, Kung CC, Liao KS, Jan JT, Ma C, Wong CH*, 2022, “Glycosite-deleted mRNA of SARS-CoV-2 spike protein as a broad-spectrum vaccine.”, Proceedings of the National Academy of Sciences of the United States of America, 119(9), e2119995119. (SCIE)
- Lee WH, Chen X, Liu IJ, Lee JH, Hu CM, Wu HC, Wang SK, Lee WH, Ma C*, 2022, “Structural basis of interleukin-17B receptor in complex with a neutralizing antibody for guiding humanization and affinity maturation”, Cell Reports, 41(4), 111555. (SCIE)
- KYA Huang, D Zhou, TK Tan, C Chen, HME Duyvesteyn, Y Zhao, HM Ginn, L Qin, P Rijal, L Schimanski, R Donat, A Harding, J Gilbert-Jaramillo, W James, JA Tree, K Buttigieg, M Carroll, S Charlton, CE Lien, MY Lin, CP Chen, SH Cheng, X Chen, TY Lin, EE Fry, J Ren, C Ma, AR Townsend, DI Stuart, 2022, “Structures and therapeutic potential of anti-RBD human monoclonal antibodies against SARS-CoV-2”, Theranostics, 12, 1-17. (SCIE)
- HY Huang, HY Liao, X Chen, SW Wang, CW Cheng, M Shahed-Al-Mahmud, YM Liu, A Mohapatra, TH Chen, JM Lo, YM Wu, HH Ma, YH Chang, HY Tsai, YC Chou, YP Hsueh, CY Tsai, PY Huang, SY Chang, TL Chao, HC Kao, YM Tsai, YH Chen, CY Wu, JT Jan, TJR Cheng, KI Lin*, C Ma* and CH Wong*, 2022, “Vaccination with SARS-CoV-2 spike protein lacking glycan shields elicits enhanced protective responses in animal models”, Science Translational Medicine, 14, eabm0899. (SCIE)
- Ko YA, Yu YH, Wu YF, Tseng YC, Chen CL, Goh KS, Liao HY, Chen TH, Cheng TR, Yang AS, Wong CH, Ma C, Lin KI, 2021, “A non-neutralizing antibody broadly protects against influenza virus infection by engaging effector cells.”, PLoS pathogens, 17(8), e1009724. (SCIE)
- Huang KYA, Tan TK, Chen TH, Huang CG, Harvey R, Hussain S, Chen CP, Harding A, Gilbert-Jaramillo J, Liu X, Knight M, Schimanski L, Shih SR, Lin YC, Cheng CY, Cheng SH, Huang YC, Lin TY, Jan JT, Ma C, James W, Daniels RS, McCauley JW, Rijal P, Townsend AR, 2021, “Breadth and function of antibody response to acute SARS-CoV-2 infection in humans”, PLOS Pathogens, 17(2), e1009352. (SCIE)
- Jan JT, Cheng TR, Juang YP, Ma HH, Wu YT, Yang WB, Cheng CW, Chen X, Chou TH, Shie JJ, Cheng WC, Chein RJ, Mao SS, Liang PH*, Ma C*, Hung SC*, Wong CH*, 2021, “Identification of existing pharmaceuticals and herbal medicines as inhibitors of SARS-CoV-2 infection.”, Proceedings of the National Academy of Sciences of the United States of America, 118(5). (SCIE)
- Liu YM, Shahed-Al-Mahmud M, Chen X, Chen TH, Liao KS, Lo JM, Wu YM, Ho MC, Wu CY, Wong CH, Jan JT, Ma C*, 2020, “A carbohydrate-binding protein from the edible Lablab beans effectively blocks the infections of influenza viruses and SARS-CoV-2”, Cell Reports, 32, 108016. (SCIE)
- Chen JR, Liu YM, Tseng YC, Ma C*, 2020, “Better influenza vaccines: an industry perspective”, Journal of Biomedical Science, 27(1), 33. (SCIE)
- Liao HY, Wang SC, Ko YA, Lin KI, Ma C, Cheng TR, Wong CH, 2020, “Chimeric hemagglutinin vaccine elicits broadly protective CD4 and CD8 T cell responses against multiple influenza strains and subtypes.”, Proceedings of the National Academy of Sciences of the United States of America, 117(30), 17757-17763. (SCIE)
- Zhou D, Duyvesteyn HME, Chen CP, Huang CG, Chen TH, Shih SR, Lin YC, Cheng CY, Cheng SH, Huang YC, Lin TY, Ma C, Huo J, Carrique L, Malinauskas T, Ruza RR, Shah PNM, Tan TK, Rijal P, Donat RF, Godwin K, Buttigieg KR, Tree JA, Radecke J, Paterson NG, Supasa P, Mongkolsapaya J, Screaton GR, Carroll MW, Gilbert-Jaramillo J, Knight ML, James W, Owens RJ, Naismith JH, Townsend AR, Fry EE, Zhao Y, Ren J, Stuart DI, Huang KYA, 2020, “Structural basis for the neutralization of SARS-CoV-2 by an antibody from a convalescent patient”, Nature Structural & Molecular Biology, 27, 950. (SCIE)
- Huang CF, Chang WH, Lee TK, Joti Y, Nishino Y, Kimura T, Suzuki A, Bessho Y, Lee TT, Chen MC, Yang SM, Hwu Y, Huang SH, Li PN, Chen P, Tseng YC, Ma C, Hsu TL, Wong CH, Tono K, Ishikawa T, Liang KS, 2020, “XFEL coherent diffraction imaging for weakly scattering particles using heterodyne interference”, AIP Advances, 10(5), 055219. (SCIE)
- Tseng YC, Wu CY, Liu ML, Chen TH, Chiang WL, Yu YH, Jan JT, Lin KI, Wong CH*, and Ma C*, 2019, “Egg-based influenza split virus vaccine with monoglycosylation induces cross-strain protection against influenza virus infections”, PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA, 116(10), 4200-4205. (SCIE)
- Chen X, Wong CH, Ma C*, 2019, “Targeting the Bacterial Transglycosylase: Antibiotic Development from a Structural Perspective”, ACS Infectious Diseases, 5(9), 1493-1504. (SCIE)
- Huang KA, Rijal P, Jiang H, Wang B, Schimanski L, Dong T, Liu YM, Chang P, Iqbal M, Wang MC, Chen Z, Song R, Huang CC, Yang JH, Qi J, Lin TY, Li A, Powell TJ, Jan JT, Ma C, Gao GF, Shi Y, Townsend AR, 2018, “Structure-function analysis of neutralizing antibodies to H7N9 influenza from naturally infected humans.”, Nature microbiology, 4(2), 306-315. (SCIE)
- Chen CL, Hsu JC, Lin CW, Wang CH, Tsai MH, Wu CY, Wong CH*, Ma C*, 2017, “Crystal Structure of a Homogeneous IgG-Fc Glycoform with the N-Glycan Designed to Maximize the Antibody Dependent Cellular Cytotoxicity.”, ACS chemical biology, 12(5), 1335-1345. (SCIE)
- Chen CP, Lin MH, Chan YT, Chen LC, Ma C*, Fischer WB*, 2016, “Membrane protein assembly: two cytoplasmic phosphorylated serine sites of Vpu from HIV-1 affect oligomerization.”, Scientific reports, 6, 28866. (SCIE)
- Lin CW, Tsai MH, Li ST, Tsai TI, Chu KC, Liu YC, Lai MY, Wu CY, Tseng YC, Shivatare SS, Wang CH, Chao P, Wang SY, Shih HW, Zeng YF, You TH, Liao JY, Tu YC, Lin YS, Chuang HY, Chen CL, Tsai CS, Huang CC, Lin NH, Ma C, Wu CY, Wong CH, 2015, “A common glycan structure on immunoglobulin G for enhancement of effector functions.”, Proceedings of the National Academy of Sciences of the United States of America, 112(34), 10611-6. (SCIE)
- Wright JD., Chu HM, Huang CH, Ma C, Chang TW, Lim C, 2015, “Structural and Physical Basis for Anti-IgE Therapy”, Scientific Reports, 5(1), 11581. (SCIE)
- Wu HH, Hwang-Verslues WW, Lee WH, Huang CK, Wei PC, Chen CL, Shew JY, Lee EY, Jeng YM, Tien YW, Ma C, Lee WH, 2015, “Targeting IL-17B-IL-17RB signaling with an anti-IL-17RB antibody blocks pancreatic cancer metastasis by silencing multiple chemokines.”, The Journal of experimental medicine, 212, 333-349. (SCIE)
- Huang CK, Yang CY, Jeng YM, Chen CL, Wu HH, Chang YC, Ma C, Kuo WH, Chang KJ, Shew JY, Lee WH, 2014, “Autocrine/paracrine mechanism of interleukin-17B receptor promotes breast tumorigenesis through NF-kappaB-mediated antiapoptotic pathway.”, Oncogene, 33, 2968-2977. (SCIE)
- Chen PC, Chuang PK, Chen CH, Chan YT, Chen JR, Lin SW, Ma C, Hsu TL, Wong CH, 2014, “Role of N-linked glycans in the interactions of recombinant HCV envelope glycoproteins with cellular receptors.”, ACS chemical biology, 9(7), 1437-1443. (SCIE)
- Kung CC, Naik MT, Wang SH, Shih HM, Chang CC, Lin LY, Chen CL, Ma C, Chang CF, Huang TH, 2014, “Structural analysis of poly-SUMO chain recognition by the RNF4-SIMs domain.”, The Biochemical journal, 462(1), 53-65. (SCIE)
- Chen JR, Yu YH, Tseng YC, Chiang WL, Chiang MF, Ko YA, Chiu YK, Ma HH, Wu CY, Jan JT, Lin KI*, Ma C*, Wong CH*, 2014, “Vaccination of monoglycosylated hemagglutinin induces cross-strain protection against influenza virus infections.”, Proceedings of the National Academy of Sciences of the United States of America, 111(7), 2476-2481. (SCIE)
- Huang CY, Shih HW, Lin LY, Tien YW, Cheng TJR, Cheng WC, Wong CH*, and Ma C*, 2012, “Crystal structure of Staphylococcus aureus transglycosylase in complex with a lipid II analog and elucidation of peptidoglycan synthesis mechanism”, Proceedings of the National Academy of Sciences of the United States of America, 109(17), 6496-6501. (SCIE)
- Shih HW, Chang YF, Li WJ, Meng FC, Huang CY, Ma C, Cheng TJ, Wong CH, Cheng WC, 2012, “Effect of the peptide moiety of Lipid II on bacterial transglycosylase.”, Angewandte Chemie-International Edition, 51(40), 10123-10126. (SCIE)
- Chen JR, Ma C*, Wong CH*, 2011, “Vaccine design of hemagglutinin glycoprotein against influenza.”, Trends in biotechnology, 29, 426-434. (SCIE)
- Sung MT, Lai YT, Huang CY, Chou LY, Shih HW, Cheng WC, Wong CH* and Ma C*, 2009, “Crystal structure of the membrane-bound bifunctional transglycosylase PBP1b from Escherichia coli”, Proceedings of the National Academy of Sciences of the United States of America, 106, 8824-8829. (SCIE)
- Wang CC, Chen JR, Tseng YC, Hsu CH, Hung YF, Chen SW, Chen CM, Khoo KH, Cheng TJ, Cheng YSE, Jan JT, Wu CY, Ma C* and Wong CH*, 2009, “Glycans on influenza hemagglutinin affect receptor binding and immune response”, Proceedings of the National Academy of Sciences of the United States of America, 106, 18137-18142. (SCIE)
- Cheng TJR, Sung MT, Liao HY, Chang YF, Chen CW, Huang CY, Chou LY, Wu YD, Chen YH, Cheng YSE, Wong CH*, Ma C* and Cheng WC*, 2008, “Domain requirement of moenomycin binding to bifunctional transglycosylases and development of high-throughput discovery of antibiotics”, Proceedings of the National Academy of Sciences of the United States of America, 105, 431-436. (SCIE)