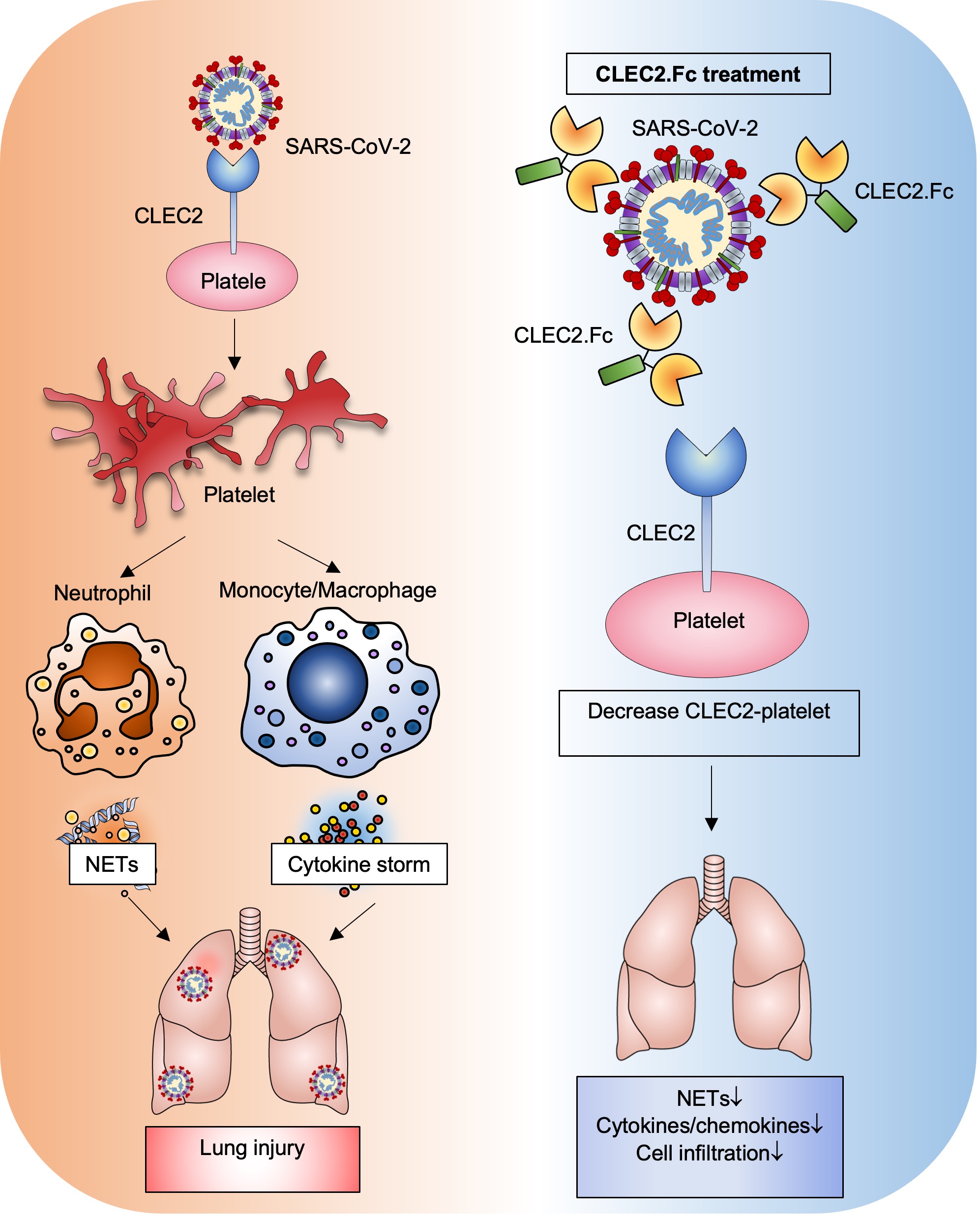
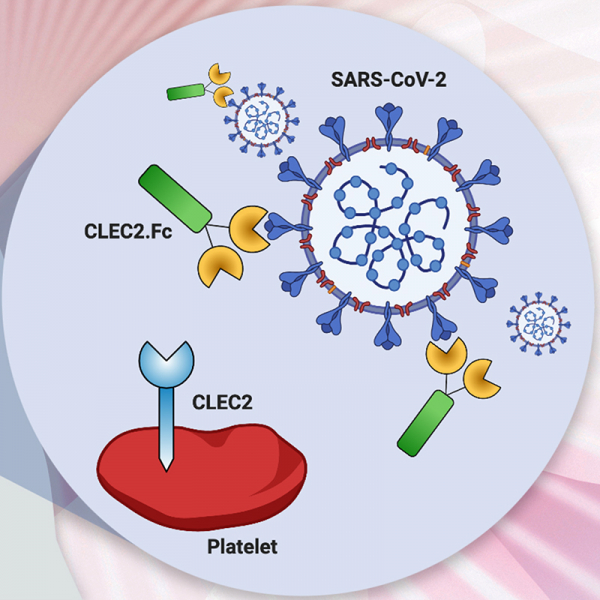
Thromboinflammation is the major cause of morbidity and mortality in COVID-19 patients, and post-mortem examination demonstrates the presence of platelet-rich thrombi and microangiopathy in visceral organs. Moreover, persistent microclots were detected in both acute COVID-19 and long COVID plasma samples. However, the molecular mechanism of SARS-CoV-2-induced thromboinflammation is still unclear. We found that the spleen tyrosine kinase (Syk)-coupled C-type lectin member 2 (CLEC2), which was highly expressed in platelets and alveolar macrophages, interacted with the receptor-binding domain (RBD) of SARS-CoV-2 spike protein (SARS-CoV-2 RBD) directly. Unlike the thread-like NETs, SARS-CoV-2-induced aggregated NET formation in the presence of wild-type (WT), but not CLEC2-deficient platelets. Furthermore, SARS-CoV-2 spike pseudotyped lentivirus was able to induce NET formation via CLEC2, indicating SARS-CoV-2 RBD engaged CLEC2 to activate platelets to enhance NET formation. Administration of CLEC2.Fc inhibited SARS-CoV-2-induced NET formation and thromboinflammation in AAV-ACE2-infected mice. Thus, CLEC2 is a novel pattern recognition receptor for SARS-CoV-2, and CLEC2.Fc and may become a promising therapeutic agent to inhibit SARS-CoV-2-induced thromboinflammation and reduced the risk of post-acute sequelae of COVID-19 (PASC) in the future.