By altering the intracellular protein modification with sugar, Dr. Kuo-I Lin’s lab made B cells in mice turn lethargic when they reach mature age and mount the antibody responses. Lin’s group has a longtime interest in pursuing the ultimate question, “what makes a B Cell THE B cell?”. They are surely one step closer to answer this question by putting the puzzle together in the latest Nature Communications article, “O-GlcNAcylation is required for B cell homeostasis and antibody responses”.
O-GlcNAcylation is a type of protein modification that is catalyzed by an enzyme called Ogt (O-GlcNAc transferase) and has been proved important during B cell activation by Lin’s group previously. Lin’s group decided to trace its significance back to the origin of the B cell, and monitor throughout the whole life span of B cells to examine if Ogt plays a role in B cell developmental stages.
The Ogt catalyzes the covalent addition of a sugar, GlcNAc, onto intracellular proteins. Aberrant O-GlcNAc modification has already been associated with diabetes, Alzheimer’s disease, cancers and stress responses in human. B cells originate from hematopoietic stem cells in bone marrow, which go through an immature stage, mature but naïve stage, and then encounter antigens and get activated in the lymphoid tissues that often form a unique B cell amplification and reaction region, referred to as germinal center. Activated B cells start making weapons called antibodies that neutralize the antigens (plasma cells), or keeping a record of the weapons for future references (memory B cells). The same mechanism applies to all mammals; therefore, in order to understand the importance of O-GlcNAc on B cells in the physiological state, they modified the Ogt gene of the mice first.
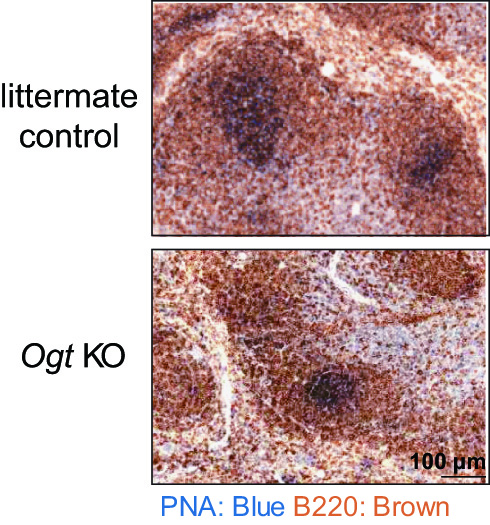 |
| Mice lacking Ogt in B cells show smaller germinal center, as revealed by less PNA+ GC B cells (blue color), after immunization. |
According to Dr. Jung-Lin Wu, one of the two first authors of this study, they spent more than half of a year to raise enough genetically modified mice for this study. The conventional Ogt deletion is embryonic lethal; therefore, to study the role of O-GlcNAcylation in B cells, the group had to generate the B-cell specific deletion of Ogt mice. Two types of Ogt knockout mice have been generated in Dr. Lin’s group: one with Ogt deficiency starting from early B cell development in bone marrow and the other lacking Ogt in the germinal center stage.
As a result, when B cells in mice have their Ogt gene removed from very early stage of their life journey, they do not only die quicker at the mature B cell stage, due to the defect in transmitting survival signal, but also lose the ability to active through B cell receptor and to form proper germinal center. Additionally, B cells deficient in Ogt in germinal center stage also fail to make the magic bullet, the antibody, as well as the memory B cells. Dr. Takashi Angata at Institute of Biological Chemistry, Academia Sinica and Dr. Pang-Hung Hsu at National Taiwan Ocean University dissected and identified the critical O-GlcNAcylated proteins in B cells.
The study provides a complete picture of how the Ogt gene empowers the B cell from the day it is born till the end of its life, enabling B cells to survive in our body and mount sufficient antibody responses. As O-GlcNAc reflects the level of nutrient status, in particular that of sugar (glucose) in our body, it appears possible that sugar level in the body may alter the B cell survival and antibody production. Further work will elucidate the roles of O-GlcNAcylation in the diseases involving B cells, which may shed light on the connection between nutrient status, metabolism and proper immune responses. The article detailed their study can be read online at: https://www.nature.com/articles/s41467-017-01677-z