O -GlcNAc is a monosaccharide spotted inside cells that has caught attentions lately. Although a very basic unit of carbohydrate, it is a big deal when noted to have shown up in the event of phosphorylation in the cell. Phosphorylation is a recognized process for messages to pass around within cells in order to carry out biological functions. Scientists began to believe O-GlcNAcylation has a role in it as well, however, the exact interactions is yet to be found out.
Dr. Kuo-I Lin has dedicated to the study of regulatory mechanisms of immune responses for a long history, she teamed up with Dr. Yu-Ju Chen from Institute of Chemistry to elucidate the role of O-GlcNAc in B cells. By analyzing various conditions of phosphorylation within B cells, they made a conclusion that the two processes happen in a timely order inside an activated B cell. By looking at the Lsp1 protein, they showed that O-GlcNAcylation helps the phosphorylation, however, the intensity of O-GlcNAcylation could ultimate cause the death of the B cell.
 Figure 1 From left: Hsin-Yi Wu, Yu-Ju Chen,Kuo-I Lin, Jung-Lin Wu
Figure 1 From left: Hsin-Yi Wu, Yu-Ju Chen,Kuo-I Lin, Jung-Lin Wu
This study is the first one to explain the intricate interplay between O-GlcNAcylation and phosphorylation in activated B cells. An article was published August 24th 2016 in Nature Communications.
B cells express B cell receptors (BCRs) on their cell membrane. BCRs are sensors in catching specific intruders, that is, the antigens. Furthermore, B cells make antibodies accordingly to protect our body. The moment when BCRs activated, a chain of reactions followed within the cell, for the sake of defense.
In the course of B cell activation, a series of messages passing started according to genomic functions. The message passing depends on protein phosphorylation at various sites on proteins. When the actions are not active as they should, B cells would not have enough power to produce proper antibodies, on the other hand, over activated B cells may cause its apoptosis or eventually leading to autoimmune diseases.
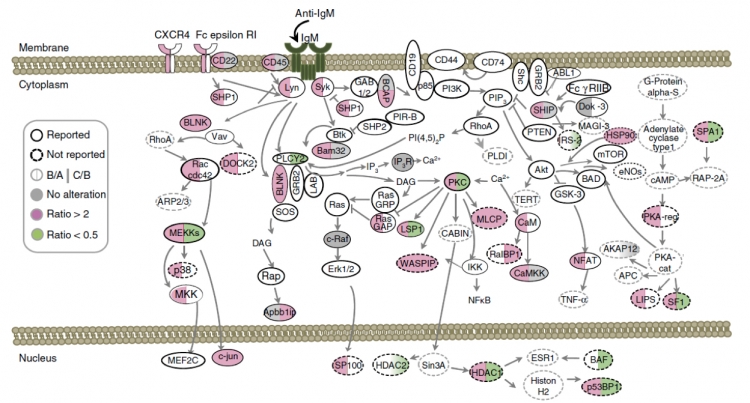 Figure 2 After B cell activation by detecting anti-IgM, a series of message passing are underway
Figure 2 After B cell activation by detecting anti-IgM, a series of message passing are underway
The phosphorylation happens on the Serine and Threonine residues, and O-GlcNAcylation is spotted on Serine and Threonine as well. Are they competitors or team players? The team decided to tackle this question.
Their approach was to obtain activated B cells at various stages of activation, and use the mass spectrometry analysis to explore it. According to Dr. Jung-Lin Wu, first author of this journal article, they extracted mouse splenic B cells, and initiate the activation in-vitro by adding anti-IgM molecule that activates BCR. Taking the frozen B cells to Dr. Hsin-Yi Wu in Dr. Chen’s lab, a series of actions started to study the amount of phosphorylation on Serine and Threonine containing peptides.
In order to clarify the glycosylation and phosphorylation interactions, they used an inhibitor of OGA that removes O-GlcNAc from conjugated proteins to detect the phosphorylated proteins relying on O-GlcNAcylation. By looking into the Lsp1 protein in B cell, when activation started, at location 209, O-GlcNAcylation is observed on Serine. Once this single sugar molecule attached to Serine, it attracts an enzyme called PKC. PKC is subsequently inducing phosphorylation at location 243 of LSP1. It turned out that the O-GlcNAcylation on Serine is a precursor for the following phosphorylation which kicks start the messaging.
On the other hand, when the phosphorylation on 243 gets too intense, it will trigger the apoptosis on the B cell, and die.
The conclusion of this research points out that for B cells, the O-GlcNAcylation is a must for follow up messaging pathways to fulfill the activated missions, yet, too much of it could simply put the life of the B cell to an end.
This study has raised an immediate interest for further study, for it has opened a door to the study of autoimmune diseases. Perhaps finding a way to balance the O-GlcNAcylation can be a new way for control.
The paper titled “Temporal regulation of Lsp1 O-GlcNAcylation and phosphorylation during apoptosis of activated B cells” can be read online at Nature Communications website.