Among many strategies in pursuit of new antibiotics for treating resistant bacterial infections, one important approach is to identify and exploit novel targets for the development of new inhibitors. A fundamental process of bacteria, bacterial cell division, is recently recognized as a novel antibiotic target, encouraging the development of new antibiotics. However, the lack of the structural information of its machinery, called the divisome, hinders the exploration of divisome-targeted antibiotics.
Membrane protein FtsB-FtsL-FtsQ (FtsBLQ) complex are essential component of divisome, which play important roles in the divisome assembling and the division-mediated peptidoglycan synthesis regulation, yet with the unclear mechanism.
In the latest publication in Nature Communications, a structure of the trimeric FtsB-FtsL-FtsQ complex was revealed for the first time, led by TIGP PhD student Ms. Hong Thuy Vy Nguyen and postdoc fellow Dr. Xiaorui Chen, from a fruitful collaboration between Dr. Che Alex Ma’s lab in the Genomics Research Center and Dr. Yu-Ling Shih’s lab in the Institute of Biological Chemistry. A structure-based model that delineates the mechanism of the regulation of peptidoglycan synthases by the FtsBLQ complex were also proposed accordingly in this study.
The research showed that FtsBLQ complex has an inverted V-shape architecture with extensive interactions at the membrane-distal region. The notable length difference between the two branches FtsBL coiled-coil and FtsQ suggests a tilted membrane insertion of the whole complex. This unique conformation of FtsBLQ facilitates the interactions with other divisome components such as its recruiter FtsK, and the downstream peptidoglycan synthase PBP1b and FtsWI in an allosteric manner, to connect and regulate the bacterial cell division process.
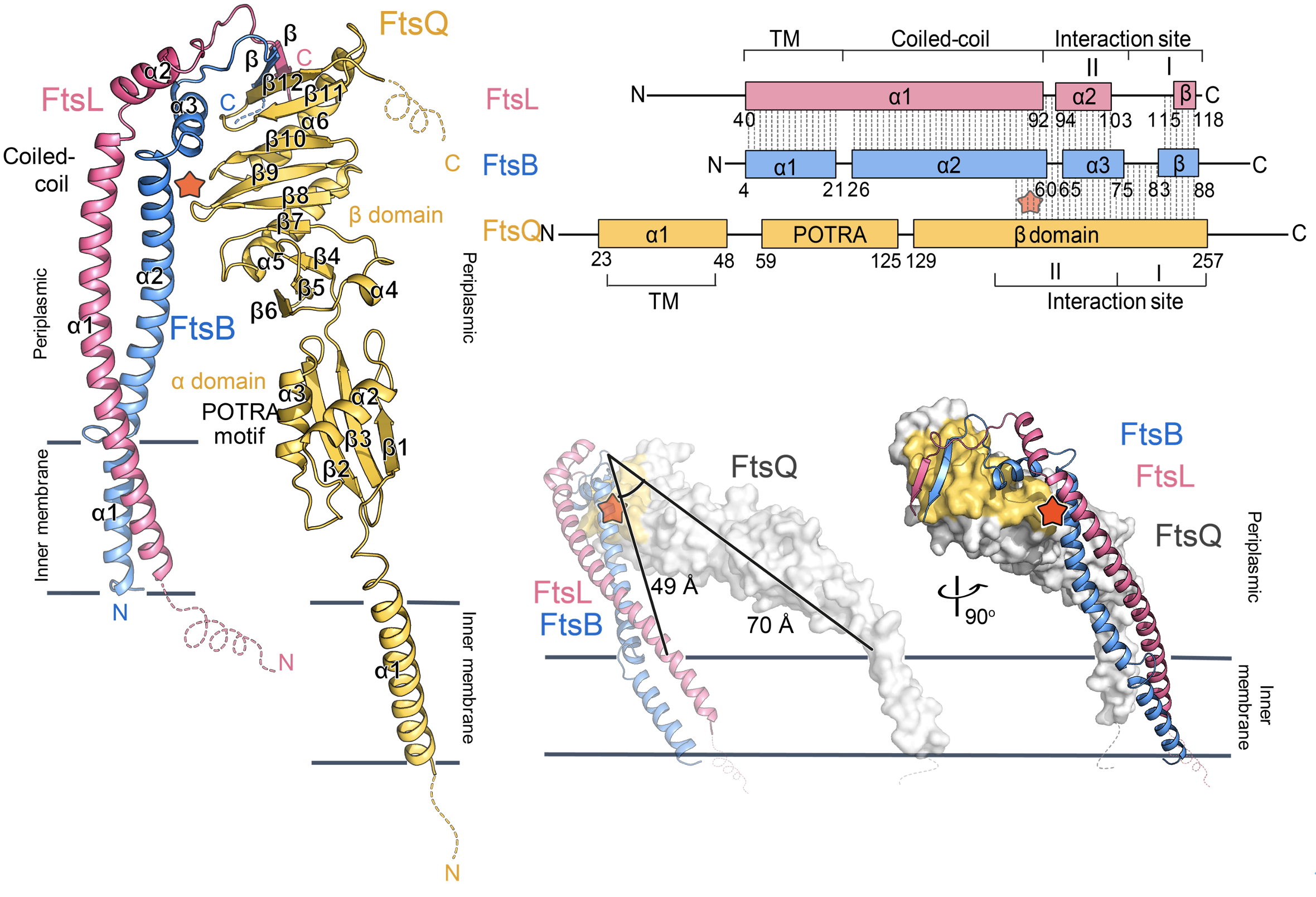 |
| Figure 1: The overall structure of FtsB-Ftsl-FtsQ complex with a titled orientation for membrane insertion. |
Two key functional domains of FtsBLQ, namely constriction control domain (CCD) and Activation FtsW-FtsI motif (AWI), are also seen for the first time in this study. Both regions are located around the linker loops between the FtsBL coiled-coil and upper region but mostly at two opposite sides. While AWI residues are surface exposed, the CCD residues, particularly FtsBE56 and FtsBD59, are buried inside, which are the only two contacts between FtsBL coiled-coil and FtsQ. The crystal structure of these mutants has also been studied, providing hints for the conformational change of FtsBL coiled-coil between different regulatory states.
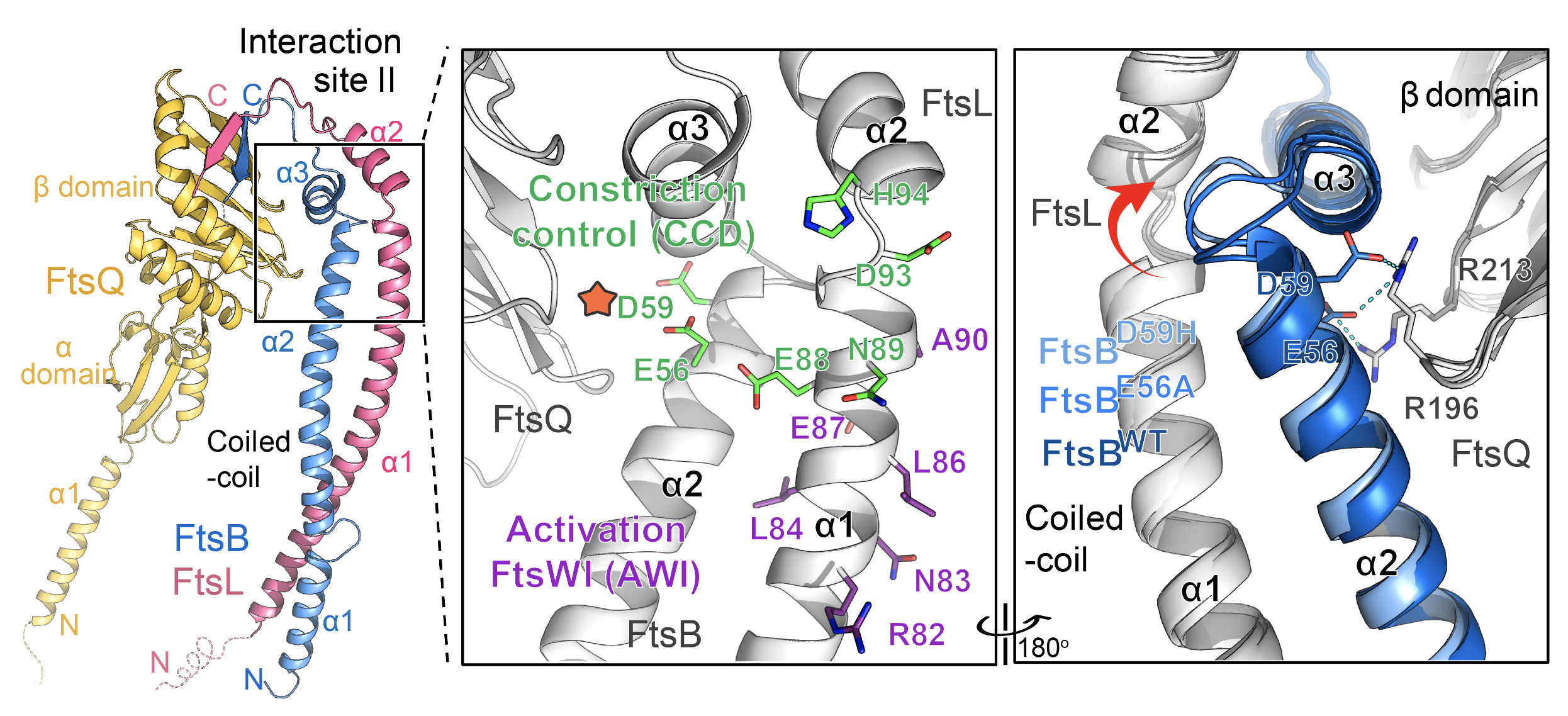 |
| Figure 2: Location of key functional residues is revealed. |
As these residues mimic the function of FtsN in the natural cell division, a structural-based model of the natural divisome-mediated peptidoglycan synthesis mechanism has also been proposed in this study.
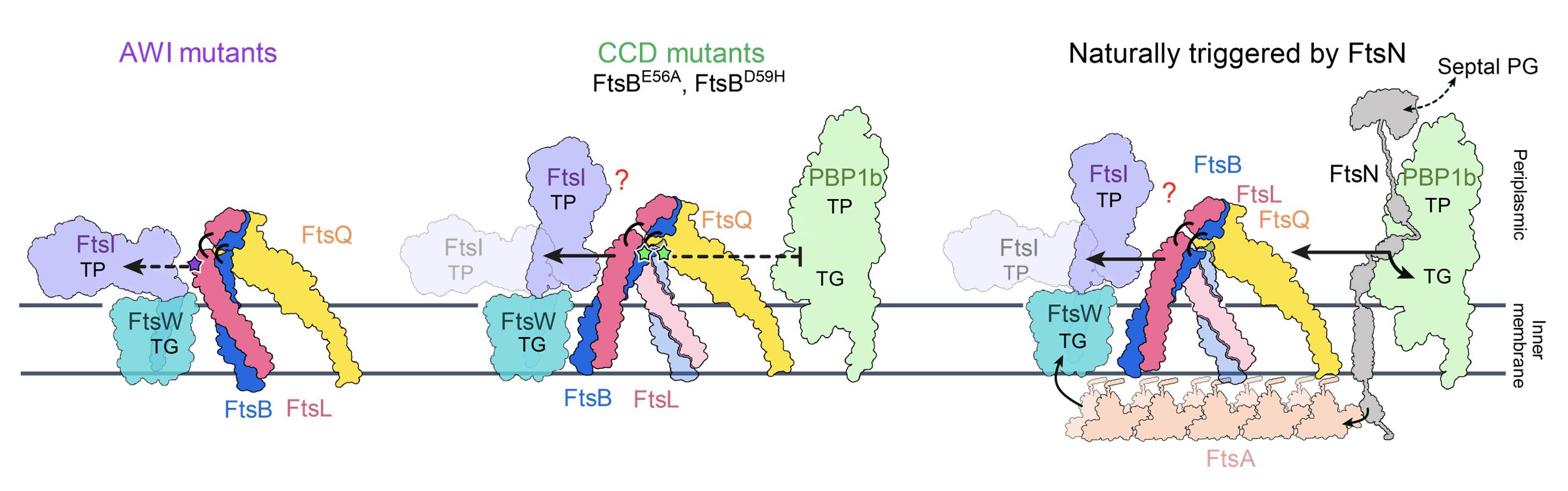 |
| Figure 3: A hypothetical model for the regulation of peptidoglycan synthases by FtsBLQ. |
These results provide significant insights not only into an understanding of the division-mediated peptidoglycan synthesis mechanism but also into inspiration for the divisome-targeted antimicrobial drug design.
A full study “Structure of the heterotrimeric membrane protein complex FtsB-FtsL-FtsQ of the bacterial divisome” can be read online at: https://www.nature.com/articles/s41467-023-37543-4