 客座講座
客座講座
EDUCATION AND POSITIONS HELD:
- B.S. & M.S., Chemistry, National Tsing Hua University, Taiwan, 1966-1972
- Ph.D., Cell and Developmental Biology, Harvard University, 1973-1977
- Postdoctoral Fellow, Center for Cancer Research, M. I. T., 1977-1980
- Supervisor of Cellular Immunology, Ortho Pharmaceutical Corp., 1980-1981
- Director of Immunology, V. P. of Research, Centocor, Inc., 1981-1985
- Professor of Molecular Virology, Baylor College of Medicine, 1986-1991
- Cofounder 1986, and V.P. of R & D, 1986-1996, Tanox, Inc., Houston
- Professor 1996-2003; Dean 1996-1999; Tsing Hua Professor of Life Science 2003-2006; College of Life Science, National Tsing Hua University
- President, Development Center for Biotechnology, Taipei, 2000-2003
- Distinguished Professor, Genomics Research Center, Academia Sinica, 2006-2015
- Distinguished Visiting Chair, Genomics Research Center, Academia Sinica, 2016-present
HONORS:
- Foundation for the Advancement of Outstanding Scholarship Award, 1997-2002
- Appointed Science and Technology Advisor of the Executive Yuan, 2002-2006
- Xolair (Chang's anti-IgE invention) approved by FDA, USA, 2003
- Appointed Tsing Hua Professor of Life Science, 2003-2006
- "Honorary Fellow Award" from American College of Allergy, Asthma, and Immunology (ACAAI), 2004
- Nature Biotechnology's shortlist of personalities who made the most significant contribution to biotech in the past 10 years. Nature Biotechnology 24, 291-300, 2006
- Xolair chosen for Prix Galien Award for outstanding innovation in R&D, UK, 2006
- "Honorary Fellow Award" from American Academy of Allergy, Asthma, and Immunology (AAAAI), 2007
- "Father of Xolair" plaque from Novartis, in Middle East Asthma and Allergy Conference, Dubai, 2012
- "Lifetime Achievement Award in Allergy" from Taiwan Academy of Pediatric Allergy, Asthma and Clinical Immunology, 2013
- TWAS(The World Academy of Sciences) Prize in Medical Sciences, 2014
RESEARCH INTERESTS:
New drug discovery and antibody engineering
The main focus of our group is to develop humanized antibody-based and immunogen-based therapeutics, which target key molecules involved in IgE-mediated allergic pathway. We are also developing new technology platforms for improved antibody engineering. One such program is to develop humanized antibody against CεmX domain in human membrane-bound IgE, for the purpose of controlling IgE-expressing B lymphocytes. CεmX, discovered by our group, is a 52 a.a. domain with a unique sequence. Anti-CεmX, if successfully developed, may be used in combination with an anti-IgE antibody, such as omalizumab (trade name Xolair), which is also derived from Dr. Chang's invention and which is approved for allergic asthma.
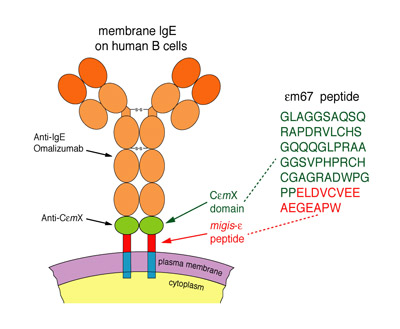
我們的研究主軸是利用抗體工程技術來開發新藥。這些以抗體為結構基礎的藥物,主要標的牽涉於IgE引致的過敏反應過程。我們也積極從事開發可提升抗體工程 的數種創新技術平台。其中一項研究計畫就是要發展人源化、高親和力,及對人體膜IgE分子內CεmX具結合特異性的抗體,以用來控制表現IgE的B淋巴細 胞。CεmX是張教授的研究群發現的;它是一含有具特異序列的52個氨基酸長的胜肽區段。如發展成功,anti-CεmX可與張教授發明的anti-IgE,如已在美國等國核准上市用於嚴重哮喘的omalizumab(商名Xolair),共同使用。